AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 
Thomson measured the weight of cathode rays and showed that they were actually a beam of particles. German scientists Eilhard Wiedemann, Heinrich Hertz and Goldstein said they were some new form of electromagnetic radiation. Scientists Crooks and Arthur Schuster said they were electrically charged atoms. 
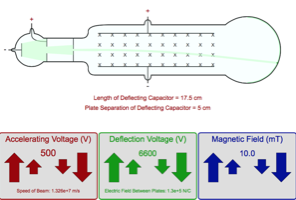
Scientists came up with two theories regarding cathode rays when they were originally discovered. Eugene Goldstein was the one who actually gave cathode rays their name. The greater part of an atom’s volume is empty space.Cathode rays were first identified by a German physicist named Johann Hittorf when he realized that something was travelling through the tube. The particle which is positively charged is called a proton. Most of the mass of the atom and all of its positive charge are contained in a small nucleus, called a nucleus. All in all, from all this we learn that the electrons are in fact the basic constituent of all the atoms. And the significant observation that he made was that the characteristics of cathode rays or electrons did not depend on the material of electrodes or the nature of the gas present in the cathode ray tube. This theory further helped physicists in understanding the structure of an atom. Thomson concluded that rays were and are basically negatively charged particles present or moving around in a set of a positive charge. So the constituents of the discharge tube were negatively charged.Īfter completing the experiment J.J. By carefully observing the places where fluorescence was observed, it was noted that the deflections were on the positive side.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed